They are in a race to save their children from a rare disease. The new drug gave them hope.

Like other children with Hunter syndrome, Cole lacks an enzyme needed to break down certain molecules. Over time, the toxins accumulate, and the genetic disorders destroy the children’s organs, including their hearts — and in many cases, their brains, leading to symptoms such as dementia. The condition, also called mucopolysaccharidosis type II, or MPS II, affects about 500 people in the US, almost all of them boys.
Experts believe the newly approved drug, an intravenous enzyme replacement therapy developed by Denali Therapeutics, will be a game-changer — especially since the current standard of care only slows down the physical aspects of the disease. Denali’s drug also targets cognitive decline.
A new drug will not reverse a relapse that has already occurred. But it can extend children’s lives and prevent many symptoms from appearing in those who catch them early.
“If we take a child, very young, and we can treat him before the injury, now the potential is almost unlimited,” said Dr. Joseph Muenzer of the Muenzer MPS Research and Treatment Center at the University of North Carolina at Chapel Hill, who sees children with Hunter syndrome and other rare mucopolysaccharide diseases.
“We don’t know how well they will do in the future, but they will do something very different than what they would have done,” he said.
Before he started regressing, Stephens’ son, Cole, was learning to read and speak in full sentences. As the disease set in, speech became difficult: He could only string a few words together, then say just one word – “Mama” – before becoming completely speechless. Despite being a teenager now, he looks like a 3-year-old, Stephens said.
The FDA’s approval of the Denali drug was a welcome surprise not only to the families of children with Hunter syndrome, but to the rare disease community as a whole. In recent months, the FDA has come under fire for rejecting a series of treatments for rare diseases, prompting patient advocates to stage a mock coffin funeral outside the agency’s headquarters and prompting an investigation by Sen. Ron Johnson, R-Wis., who, before Denali’s approval, accused the FDA of “not looking at the treatment”.
In an email to NBC News, the FDA said the number of approvals and denials under this administration is “consistent with historical data over the past decade.” Pointed to the statement of FDA Commissioner Dr. Marty Makary, who called Denali’s approval “a historic day for children and their families battling Hunter’s disease.” He added, “We will continue to do everything we can to speed up the treatment of rare diseases.”
Those who have watched children suffer from Hunter syndrome and other rare diseases hope so, including Muenzer, who was the lead investigator in the Denali case.
“These are serious diseases,” Muenzer said. Just because they are a minority doesn’t mean we should ignore them.”
Denali’s drug, called Avlayah, is the first FDA-approved treatment in the US in 20 years for Hunter syndrome – and the first to penetrate the blood-brain barrier, allowing it to stop the neurologic complications of the disease.
Experts like Muenzer believe that Avlayah has the potential to increase life expectancy based on promising data from a clinical trial that showed that after 24 weeks, the levels of the main biomarker in the cerebrospinal fluid associated with the disease decreased significantly, 93% of the pediatric participants had levels comparable to people without Hunter syndrome.
For many families of children with this condition, the drug’s approval means more than just an exciting new treatment. It also meant realizing the importance of their children.
In New Berlin, Wisconsin, 6-year-old Roran Jaskulski was diagnosed with Hunter syndrome when he was 4 years old. He hasn’t been able to speak, and his mother, Kylie Jaskulski, said she worries that because Roran can’t talk, strangers might ask if he has anything to offer others — a heart-wrenching trauma that affects her child’s sleep and constipation. he runs to school happily every day.
“He brings so much joy,” Jaskulski said. “He brings joy and peace to everyone he comes in contact with.”
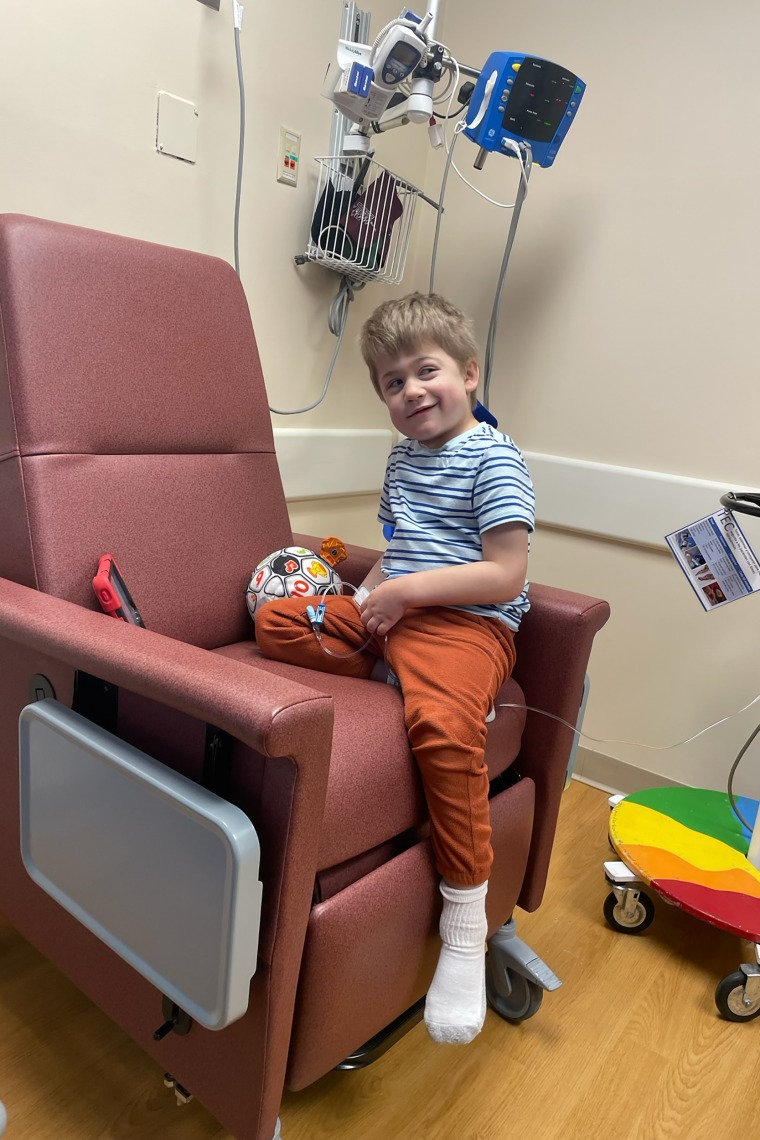
Jaskulski said at first, getting Roran’s diagnosis felt like the worst day of his life. But as time went on, he realized that his powerlessness to prevent his son from deteriorating was even more painful.
If his insurance approves Roran’s Denali drug, he said, “maybe I don’t have to wait and see.”
Like other children with Hunter syndrome, Roran receives a weekly infusion of the current standard of care, a drug called Elaprase that was approved by the FDA in 2006. The infusions help stabilize his physical decline, although not completely: In recent months, Jaskulski has noticed Roran developing weakness and pain on the left side of his body, which affects his mobility. He is also hard of hearing.
A coalition of states from California to Rhode Island includes Hunter syndrome in newborn screening, and more states are expected to begin screening for it at birth in the future. That means children will have a better chance of preventing mental retardation if they take the new drug early, doctors say.
Most children with Hunter syndrome have a severe form of the disease, living between 10 and 20 years. Those with the non-neurological form of Hunter syndrome, which does not affect the brain as much, can live into adulthood, although they still face ongoing physical problems, primarily with their airways and heart.

The prospect of a new treatment is exciting for the families of children at both ends of the disease. In Newkirk, Oklahoma, Christina Coldwell’s 3-year-old granddaughter Kashton Estes has Hunter syndrome without cognitive problems, and is receiving an infusion of Elaprase, which she refers to as “getting her Spider-Man juice.” The treatment was a success: Coldwell said the little boy is “talking up a storm,” running, jumping and riding a bike.
Still, you can’t wait to get Kashton in the new Denali car. Two of her cousins also have the genetic disease, Coldwell said, and the family wants to prevent future health problems for all three children.
“We are not asking for much: Just give us medicine to keep our children alive,” she said.
When asked about insurance coverage for its drug — which has a list price of $5,200 per 150-milligram bottle — Denali Therapeutics said it has had “constructive” discussions with payers and said enabling Hunter syndrome families to have immediate access to Avlayah is a “priority.” It also said that it is looking to expand the clinical evidence of this drug in adults, because at the moment, it is indicated only in children’s patients. Denali also said it intends to apply its blood-blocking technology to a variety of other conditions, including other neurodegenerative diseases.
Stephens, the mother of 15-year-old Cole, can’t wait to start her son on medicine. He dedicated his life to helping not only Cole, but others like him: In 2022, he became the executive director of the Muenzer MPS research and treatment center at UNC-Chapel Hill.
When news broke last week that the FDA had granted fast-track approval for the new drug, Stephens ran to the medical center to tell patients and staff. Everyone lied and cried.
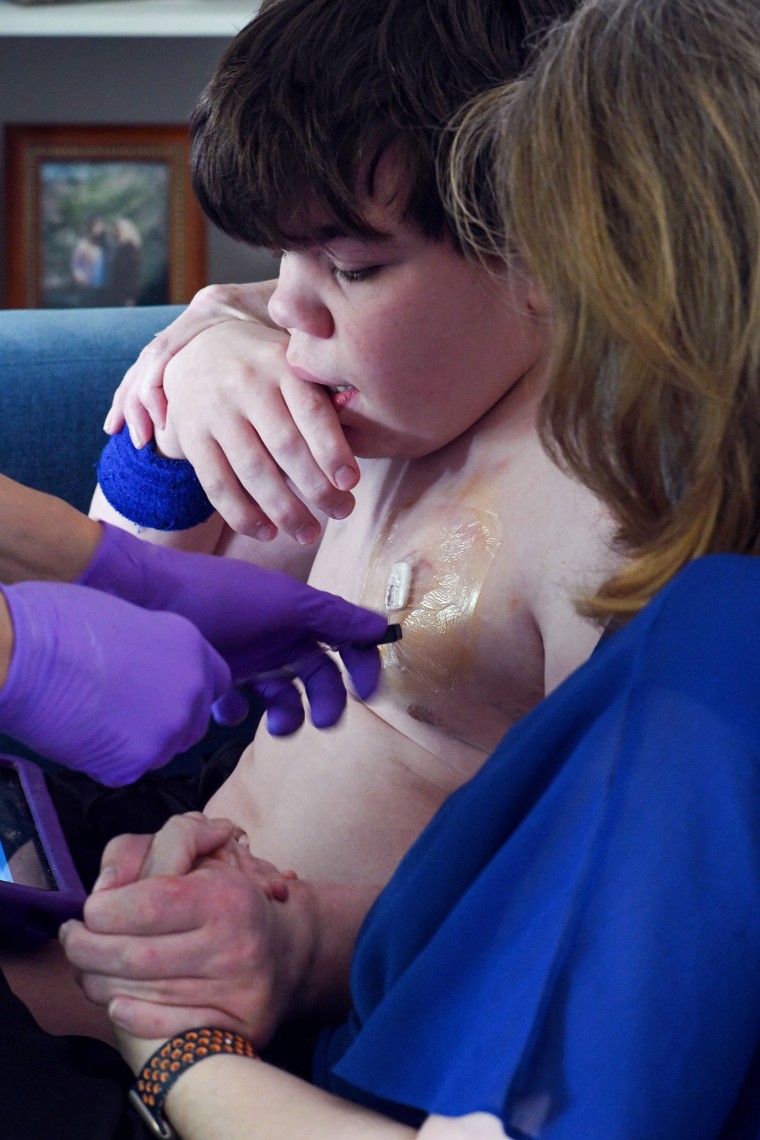
Stephens knows that Denali’s medicine cannot reverse the regression that Cole already has. It won’t enable him to go to college or get a job. But he said he is still incredibly grateful.
“My hope is that he will remain stable,” Stephens said. “Being stable with a progressive disease is a win.”


